NMPA (Class III)
Highest Safety Classification in China
Provincial-level science and technology administrative department
The Center operates under the official Charter of Nanjing Engineering Research Center for Anti-AMR Nanomaterials. For legal and financial stability, we are supported by NMS Technologies Co., Ltd., which provides the industrial infrastructure.
Led by Prof. Huining Xiao (Fellow of the Canadian Academy of Engineering), driving biodegradable and physical antimicrobial innovations.
Guided by top academicians Dr. Zhong Nanshan and Dr. Guo Yinglu.

Unlike chemical antibiotics that interfere with metabolic pathways (leading to resistance), our core technology utilizes a positively charged nano-film. Confirmed by the University of New Brunswick, the active ingredient maintains a stable Zeta Potential of +60mV .
The Effect:This strong electrostatic field physically attracts negatively charged bacteria and ruptures their cell walls upon contact.
Learn More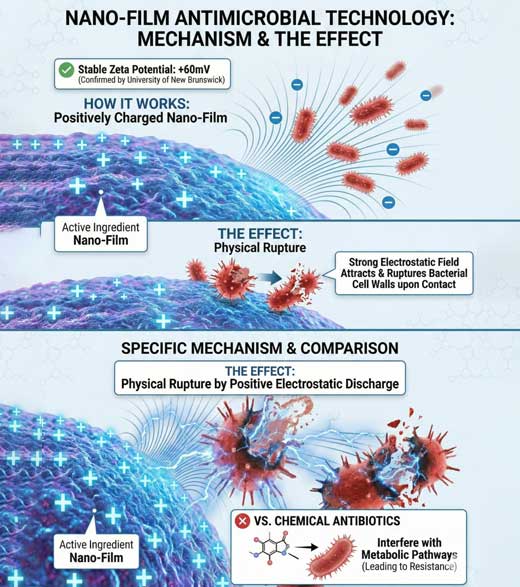

Our "Physical Antimicrobial Method" is secured by invention patents in major global economies



Massive Clinical Adoption:Over 1,000 academic papers have been published in the CNKI database validating the efficacy of this technology across diverse clinical departments.
Learn More

The "One-Glass Method" Our physical antimicrobial technology has revolutionized clinical diagnostics. By using physical antimicrobial spray to collect bodily fluids, we developed the "One-Glass test" for prostatitis diagnosis. Supported by rigorous animal and clinical trials, it replaces the international standard "Meares–Stamey 4-glass test," significantly reducing false-positive/negative misdiagnoses and lowering patient costs.
Learn MoreFor high efficacy, our physical therapy has been granted the "Strongest Recommendation" by the official Chinese Urology and Andrology Diagnosis and Treatment Guidelines.
Learn More



Curing superbug infections and preventing biofilm-related diseases without antibiotics.
Curing superbug infections and preventing biofilm-related diseases without antibiotics.
Offering a viable alternative to veterinary antibiotics. We conduct non-drug infection models and treat animal enteritis, halting the spread of AMR in livestock.
Pioneering physical anti-biofouling coatings for ships and vessels, protecting marine environments from toxic chemical pollution.
Developing biodegradable antimicrobial agricultural films and food preservation packaging materials to ensure global food safety.
For over two decades, we have actively responded to global public health emergencies, provided humanitarian aid during natural disasters, and championed health equity for vulnerable populations in underdeveloped regions.

Recognized in the Fight Against SARS During the 2003 SARS outbreak, our physical antimicrobial technology was officially designated as a "High-Tech Product for SARS Prevention" by the Chinese Ministry of Science & Technology.
• Impact: Provided critical infection control barriers for frontline healthcare workers. This established our 20-year historical legacy in national epidemic control.
The "Western China Medical Aid Tour" In collaboration with the Chinese Medical Association, we organized continuous medical aid missions to remote and resource-limited regions, including Tibet (2005), Gansu (2006), Xinjiang (2007), and Inner Mongolia (2008).
• Impact: Trained local doctors on non-antibiotic infection control, bridging the healthcare gap. This monumental effort is officially recorded in the History of Chinese Urology.


Wenchuan Earthquake Humanitarian Aid In the wake of the devastating 2008 Wenchuan Earthquake, we immediately mobilized emergency medical supplies to the disaster zones.
• Impact: Delivered crucial humanitarian aid to prevent secondary, post-disaster infections in temporary shelters and field hospitals.
Poverty Alleviation for Mountainous Schools To protect vulnerable children from infectious diseases, we launched a targeted health and medical donation initiative for Jiarong Middle School in the poverty-stricken mountainous area of Guizhou Province.
• Global Alignment: This action strictly aligns with the UN Sustainable Development Goals (SDG 1: No Poverty & SDG 3: Good Health and Well-being).

